Pharmaceutical Safety Assessment Market 2026: Compliance and Risk Evaluation Trends
The Business Research Company’s Pharmaceutical Safety Assessment Global Market Report 2026 - Market Size, Trends, And Global Forecast 2026-2035
LONDON, GREATER LONDON, UNITED KINGDOM, March 6, 2026 /EINPresswire.com/ -- The pharmaceutical safety assessment market is dominated by a mix of global contract research organizations, specialized toxicology service providers, and integrated life sciences companies. Organizations are focusing on advanced in vitro and in vivo toxicology models, predictive computational toxicology platforms, biomarker-based safety profiling, and GLP-compliant laboratory infrastructure to strengthen market presence and ensure regulatory adherence. Emphasis on regulatory compliance with international safety guidelines, early-stage risk identification, data integrity, and integration of digital data management and analytics systems remains central to competitive positioning. Understanding the competitive landscape is essential for stakeholders seeking growth opportunities, technological innovation, and strategic collaborations within the evolving drug development and preclinical safety evaluation ecosystem.
Which Market Player Is Leading the Pharmaceutical Safety Assessment Market?
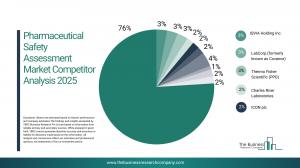
• According to our research, IQVIA Holding Inc. led global sales in 2024 with a 3% market share. The company’s research and development solutions, which is directly involved in the pharmaceutical safety assessment market, provides comprehensive preclinical and clinical safety evaluation services, including toxicology studies, pharmacovigilance, risk management, regulatory consulting, and advanced analytics that support drug development, regulatory submissions, and global compliance requirements.
Who Are The Major Players In The Pharmaceutical Safety Assessment Market?
Major companies operating in the pharmaceutical safety assessment market are IQVIA Holdings Inc., Labcorp (formerly known as Covance), Thermo Fisher Scientific Inc. (PPD), Charles River Laboratories, ICON plc, Parexel International Corporation, Syneos Health, Eurofins Scientific SE, WuXi Biologics Inc., SGS SA, Medpace Holdings Inc., Fujifilm Corporation (Fujifilm Wako), Pharmaron Beijing Co. Ltd., Inotiv Inc. (including Envigo), Evotec SE (Cyprotex), Intertek Group plc, Altasciences Inc., Aragen Life Sciences Ltd., CTI Clinical Trial and Consulting Services Inc., ChemDiv Inc., Frontage Laboratories Inc., Shanghai Medicilon Inc., Biotoxtech Co. Ltd., BioIVT LLC, Accelera Srl.
How Concentrated Is The Pharmaceutical Safety Assessment Market?
The market is moderately fragmented, with the top 10 players accounting for 24% of total market revenue in 2024. This level of concentration reflects moderate to high technological and regulatory entry barriers, driven by stringent global regulatory requirements, adherence to Good Laboratory Practice (GLP) standards, increasing complexity of toxicology and safety pharmacology studies, and the need for advanced analytical capabilities and validated data systems to ensure reliable, reproducible, and compliant safety assessment across preclinical and clinical development stages. Leading players such as IQVIA Holdings Inc., Labcorp (formerly known as Covance), Thermo Fisher Scientific Inc. (PPD), Charles River Laboratories International Inc., ICON plc, Parexel International Corporation, Syneos Health Inc., Eurofins Scientific SE, WuXi Biologics (Cayman) Inc., SGS SA hold notable market shares through comprehensive safety assessment service portfolios, long-term pharmaceutical and biotechnology partnerships, global laboratory networks, and continuous innovation in predictive toxicology and regulatory consulting solutions. As demand for integrated preclinical and clinical safety evaluation, biomarker-based risk assessment, and data-driven pharmacovigilance solutions increases, strategic collaborations, service expansion, and geographic diversification are expected to further strengthen the competitive positioning of these leading companies in the market.
• Leading companies include:
o IQVIA Holdings Inc. (3%)
o Labcorp (formerly known as Covance) (3%)
o Thermo Fisher Scientific Inc. (PPD) (3%)
o Charles River Laboratories International Inc. (2%)
o ICON plc (2%)
o Parexel International Corporation (2%)
o Syneos Health Inc. (2%)
o Eurofins Scientific SE (2%)
o WuXi Biologics (Cayman) Inc. (2%)
o SGS SA (2%)
Request A Free Sample Of The Pharmaceutical Safety Assessment Market Report:
https://www.thebusinessresearchcompany.com/sample_request?id=29147&type=smp&utm_source=EINPresswire&utm_medium=Paid&utm_campaign=Mar_PR
Who Are The Key Raw Material Suppliers In The Pharmaceutical Safety Assessment Market?
• Major raw materials suppliers in the pharmaceutical safety assessment market include Thermo Fisher Scientific Inc., Merck KGaA, Agilent Technologies Inc., Charles River Laboratories International Inc., Lonza Group Ltd., Bio-Rad Laboratories Inc., PerkinElmer Inc., QIAGEN N.V., Illumina Inc., GE Healthcare, Eppendorf SE, Corning Incorporated, Bruker Corporation, Waters Corporation, Promega Corporation, Shimadzu Corporation, Sartorius AG, Tecan Group Ltd., Beckman Coulter Inc. (Danaher), Abcam plc, Sigma-Aldrich (Merck), Takara Bio Inc., BD Biosciences (Becton Dickinson and Company), Enzo Life Sciences Inc., BioLegend Inc.
Who Are The Major Wholesalers And Distributors In The Pharmaceutical Safety Assessment Market?
• Major wholesalers or distributors in the pharmaceutical safety assessment market include Avantor Inc., VWR International (Avantor), McKesson Corporation, Cardinal Health Inc., Medline Industries Inc., Henry Schein Inc., Cole-Parmer Instrument Company LLC, Daigger Scientific Inc., Labnet International Inc., Greiner Bio-One International GmbH, Bio-Techne Corporation, GenScript Biotech Corporation, Santa Cruz Biotechnology Inc., Cayman Chemical Company, Vector Laboratories Inc., BioVision Inc., OriGene Technologies Inc., STEMCELL Technologies Inc.
Who Are The Major End Users Of The Pharmaceutical Safety Assessment Market?
• Major end users in the pharmaceutical safety assessment market include Pfizer Inc., Novartis International AG, Johnson & Johnson, Roche Holding AG, AstraZeneca plc, Sanofi S.A., GlaxoSmithKline plc (GSK), Amgen Inc., Regeneron Pharmaceuticals Inc., Moderna Inc., BioNTech SE, Vertex Pharmaceuticals Incorporated, Eli Lilly and Company, Takeda Pharmaceutical Company Limited, Bristol-Myers Squibb Company, Merck & Co. Inc., AbbVie Inc., Boehringer Ingelheim International GmbH, Bayer AG, CSL Behring, CRISPR Therapeutics AG, Alnylam Pharmaceuticals Inc., Gilead Sciences Inc.
What Are the Major Competitive Trends In The Market?
• Artificial intelligence-powered decision support platforms are transforming the pharmaceutical safety assessment market by enhancing the accuracy of toxicity prediction, accelerating pathology workflows, and enabling data-driven risk evaluation in preclinical studies.
• Example: In April 2024, Charles River Laboratories launched the Patholytix Foresight platform, an AI-powered decision support tool designed to modernize toxicologic pathology in pharmaceutical safety assessment.
• Its advanced AI algorithms rapidly analyze complex preclinical pathology data to detect tissue abnormalities with high precision, enabling earlier identification of potential safety risks and reducing subjectivity in interpretation. Its cloud-based architecture supports global collaboration and scalable deployment, while significantly reducing slide review time, improving data consistency, and enhancing overall efficiency and reliability in pharmaceutical safety assessment processes.
Which Strategies Are Companies Adopting To Stay Ahead?
• Leveraging Innovative Pharmacovigilance Approaches To Minimize Redundancy And Enhance Drug Safety Monitoring
• Advancing Comprehensive Risk Management Strategies Across The Drug Lifecycle
• Enhancing Drug Residue Toolboxes To Strengthen Safety Standards And Support Regulatory Compliance
• Cross-Species Cardiac Research Models Improve Predictive Safety Assessment And Translational Drug Development Outcomes
Access The Detailed Pharmaceutical Safety Assessment Report Here:
https://www.thebusinessresearchcompany.com/report/pharmaceutical-safety-assessment-global-market-report?utm_source=EINPresswire&utm_medium=Paid&utm_campaign=Mar_PR
Learn More About The Business Research Company
The Business Research Company (www.thebusinessresearchcompany.com) is a leading market intelligence firm renowned for its expertise in company, market, and consumer research. We have published over 17,500 reports across 27 industries and 60+ geographies. Our research is powered by 1,500,000 datasets, extensive secondary research, and exclusive insights from interviews with industry leaders.
We provide continuous and custom research services, offering a range of specialized packages tailored to your needs, including Market Entry Research Package, Competitor Tracking Package, Supplier & Distributor Package and much more.
Disclaimer: Please note that the findings, conclusions and recommendations that TBRC Business Research Pvt Ltd delivers are based on information gathered in good faith from both primary and secondary sources, whose accuracy we are not always in a position to guarantee. As such TBRC Business Research Pvt Ltd can accept no liability whatever for actions taken based on any information that may subsequently prove to be incorrect. Analysis and findings included in TBRC reports and presentations are our estimates, opinions and are not intended as statements of fact or investment guidance.
Contact Us:
The Business Research Company
Americas +1 310-496-7795
Europe +44 7882 955267
Asia & Others +44 7882 955267 & +91 8897263534
Email: info@tbrc.info
Follow Us On:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
Oliver Guirdham
The Business Research Company
+44 7882 955267
info@tbrc.info
Visit us on social media:
LinkedIn
Facebook
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.